Find answers and solutions to common questions for the Bluelab pH Pen, and the Bluelab Soil pH Pen
In this guide:
General questions
My pH product won’t calibrate, what should I do?
The first step is to make sure the glass tip of the probe is both well hydrated and clean.
If, after both hydrating and cleaning, your pH pen still won’t calibrate, it may be at the end of its life. Please reach out to our support team if you have further questions.
My pH reading is stuck on 7 pH, what should I do?
Inspect the glassware for damage. If the pen reading is stuck on 7 pH (or close to that), it is likely the glassware is damaged. The pH pen likely needs replacing.
My pH probe has accidentally dried out, what should I do?
The probe must be rehydrated for 24 hours in KCI storage solution (never use RO, distilled or deionised water). Rinse the probe in fresh tap water, and soak the probe in fresh pH 4.0 Calibration Solution for at least 10 minutes. Calibrate the probe to two points following the calibration article, to check the probe hasn’t suffered permanent damage.
Ensure the sponge in the pH Pen storage cap contains KCl Storage Solution, or fill the pH Probe cap with enough KCl to cover the sensor so the probes remain hydrated.There is a salt crust on my probe – why?
Both the pH pen/probe and the storage cap contain KCl (Potassium Chloride) solution. The result of a small level of evaporation of KCl is the formation of a salt crust as KCl is a type of salt.
This salt crust is normal and will not harm the probe or meter. Gently wash the salt off under fresh running water. It is essential to ensure that your probe has sufficient KCl in the storage cap at all times to remain hydrated.
How long will my Bluelab pH Pen last?
Bluelab's pH products are charged with reference material which expends over time and use. The average life span of a pH pen is approximately 18 months. Good care will help to maintain your pen. Follow our guides to proper hydration, regular cleaning and calibration.
The pH pen has a 1-year warranty so if you encounter any issues with your current unit, please feel free to reach out to us for support.
What is KCl Storage Solution?
Our Bluelab pH Probe KCl (Potassium Chloride) Storage Solution is a storage solution designed specifically for use with Bluelab pH Pens or pH Probes.
Storage solution is used to ensure a pH probe is kept in ideal conditions during storage, while the probe is not in use. Read more about KCl solution.
Understanding error messages
| Error code | Reason | Correction |
|
Temperature under range
|
Calibration solutions may be contaminated | Always use fresh calibration solutions |
| Probe not hydrated or clean | Follow the cleaning and calibration steps | |
| Probe damaged | Replace the pH pen | |
|
|
Wrong calibration sequence | Calibrate to pH 7 first, then to pH 4 |
| Probe damaged | Replace the pH pen | |
|
pH calibration failed |
Calibration solutions may be contaminated | Always use fresh calibration solutions |
| Probe not hydrated or clean | Follow the cleaning and calibration steps | |
| Probe damaged. | Replace the pH pen | |
|
Hardware error |
The pH probe may be damaged | Check for broken glass on the tip of the pen’s pH probe. Consider replacing your pH pen. |
Troubleshooting
| Trouble | Reason | Correction |
| Drift – readings slowly varying | Glassware not clean | Clean glassware, hydrate, then calibrate |
| Wick contaminated or blocked | Soak probe in KCl storage solution for 24 hours and retest. Don't measure proteins or oils with the pH pen. You may need to replace the pH pen. | |
| Glassware aged | Replace the pH pen | |
| Unsuccessful calibration | Glassware not clean | Clean glassware |
| Glassware aged (glassware won't clean) | Replace the pH pen | |
| Probe not hydrated | Soak the probe in KCl solution for 24 hours and retest | |
| Noisy – readings jumping around | Contact zone not immersed correctly | Lower pen into solution at least 2cm/1" |
| Reading stuck on 7 pH | Broken glassware | Replace the pH pen |
| Incorrect sample reading following successful calibration | Wick blocked |
Soak probe in KCl storage solution for 24 hours and retest. Don't measure proteins or oils with the pH pen. |
Additional information regarding the pH Pen or pH Pen Soil can be found on the manual:
pH Soil pH manual (English)
Still can’t find what you're looking for? Contact Support

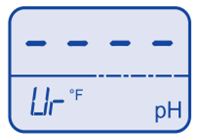
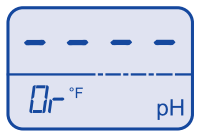 Temperature over range
Temperature over range 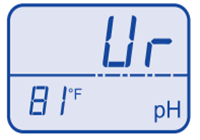 pH under range
pH under range 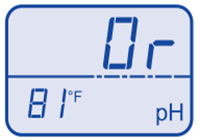 pH over range
pH over range 
