An introduction to measuring pH direct in the root zone of your plants
An introduction to measuring pH in soil, preparation and factors affecting soil pH
In this article
An introduction to pH and soil
Precautions to measuring pH in soil
Factors affecting pH in soil or media
An introduction to pH and soil
pH is the measurement of the hydrogen ion concentration (H+) — acidity, and its opposite, alkalinity.
- Neutral pH is 7.0 pH
- Acidity measures below 7.0 pH
- Alkalinity measures above 7.0 pH
In soils or growing media, pH influences the availability of nutrients and the presence of microorganisms in soil.
Certain plants require a particular pH range to enable the required nutrients to be consistently available to the plant. If the solution is too acidic or too alkaline it can cause “lockup” – a situation that restricts certain elements essential for growth from being absorbed by the root structure. This, in turn, reduces plant health and performance.
Plant nutrient deficiencies can lead to crop failure. For example:
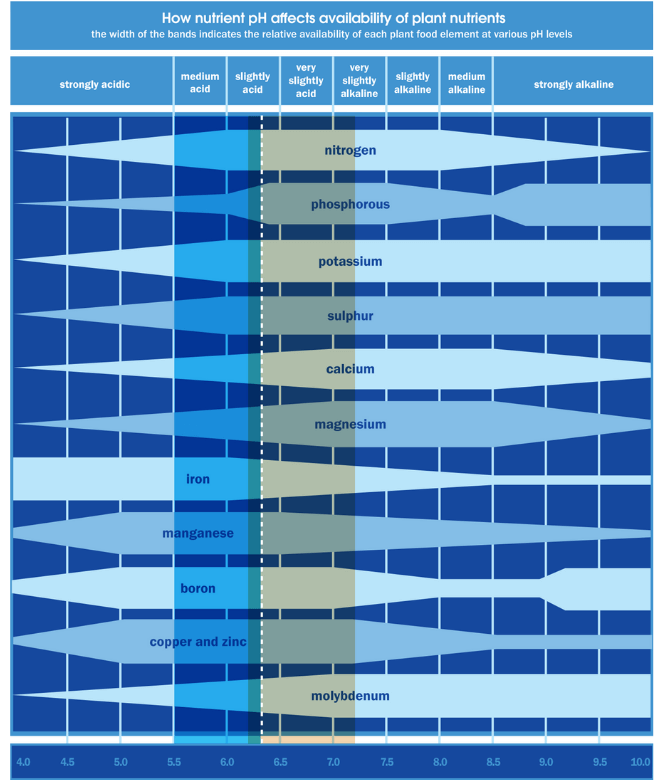
- Low soil pH causes aluminium and manganese toxicity in plants and reduces the availability of soil phosphorus.
- High soil pH also reduces soil phosphorus availability and reduces the availability of micronutrients such as zinc and boron to plants.
The chart below shows how nutrient pH levels influence the uptake of certain elements.
Precautions to measuring pH in soil
Taking pH measurements of soil with an electric meter is indicative rather than absolute.
The following factors are outside the control of any soil pH meter, so to minimise their effect on the accuracy of the pH measurement, consider the following.
Moisture level/raw water
If the sample you are wishing to measure is dry, add RO water or distilled water to moisten. Ideally wait 24 hours before you take a measurement.
Note: If you add tap water to your soil, you will influence the soil pH reading based on the pH of the tap water.
Calibration of the Soil pH Pen and cleanliness of the soil probe tip
Calibrating the Soil pH Pen at least monthly will ensure accurate readings. Cleaning the soil residue from the probe tip and storing the Soil pH Pen in a clean moist state will provide reliable readings as well as prolonging the pens life.
Read more about how to calibrate the Bluelab Soil pH Pen
Sample selection
For field testing, remove the top 5 - 10 cm / 2 - 4” of the top of the soil. Samples are taken
approximately 15 - 20 cm / 6 - 8” down into the substrate and from various areas, then an average of the readings is used.
For container grown plants, it is recommended to check the pH level of the substrate prior to planting.
Factors affecting pH in soil or media
There are some factors outside your control that affect soil pH. Here are 6 you should be aware of.
1. Soil type
Soils formed under high rainfall conditions (e.g. Eastern USA) are more acidic than those formed under dry conditions (e.g. Western USA).
2. Growth stage of the plant
Uptake and requirements of particular elements change as the plant progresses through it’s growing cycle. Recording pH level data to create a history is valuable.
3. Applications and types of fertilisers
Applications and types of fertilizers can alter the pH level significantly. The time at which you take the reading is important. Evaluate the brand of fertilizer to see if it is altering the pH in the wrong direction.
4. Sprays
As sprays can soak into the soil/media, a change to the pH level could result.
5. Soil/media temperature
High temperature soils may have a high concentration of CO2. The higher the concentration of carbon dioxide pressure results in more carbonic acid which lowers pH.
6. pH range for soil crops
The recommended pH range for soil crops is 6.2 - 7.2, but this is plant-specific.
Still can’t find what you're looking for? Contact Support
%20copy-1.png?width=200&height=58&name=Bluelab-logo__Trans_RGB-medium%20(2)%20copy-1.png)